Jun 22, 2021
1 min, 21 secs
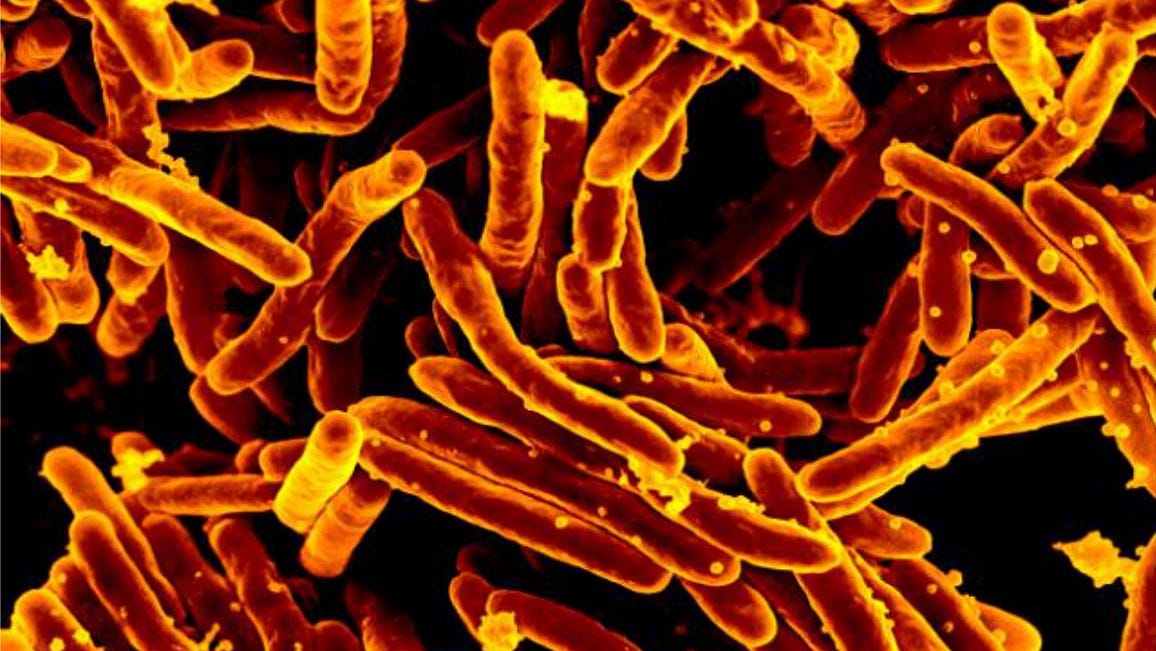
Delaware and federal health officials are investigating reports of patients contracting tuberculosis following certain spinal surgeries, part of an outbreak of more than 100 cases nationwide. .Richard Williams, 74, of Bear, became one of the first of these patients to file a lawsuit against Aziyo Biologics, a regenerative medicine company that created the product use in the surgery.
Health officials do not believe any other units were infected, and all of the unused products have been put aside.
As of June 15, health care providers across the country have contacted 105 patients who had surgery using this product.The CDC recommends that all patients who received this product immediately start full treatment for TB, even if they do not show any symptoms.It was the result of "a customer complaint from one hospital that initially reported post-surgical infection in 7 of the 23 patients that have received FiberCel from this Donor Lot," according to an FDA notice
Williams, the lawsuit alleges, was one of 23 Delaware patients who received this infected productIt forced him to undergo a “painful and complicated revision surgery†to remove the infected product and insert a new bone product, according to the lawsuit.Â
In May, ChristianaCare “identified a small number of patients who tested positive for tuberculosis infection following surgery involving a specific product,†according to a hospital spokesmanThe health system said it immediately contacted the health department and FDA, and also launched its own investigation. Mary Fenimore, a spokeswoman for the Division of Public Health, said the division was notified in late May by a health care system of a "cluster of patients who developed Mycobacterium tuberculosis following surgery involving a specific product."