Following encouraging results, they administered the drug to a patient with ALS.
Compared to normal mice, mice with a mutated FUS gene had higher levels of insoluble FUS and other ALS-related proteins in their brains and spinal cords.Mice with higher doses of mutant FUS in motor neurons experienced rapid neurodegeneration that began early in life, much like FUS-ALS patients.
In mice, injecting a single dose of the drug into the ventricles, fluid-filled spaces surrounding the brain, delayed the onset of inflammation and motor neuron degeneration by six months.The drug also knocked down levels of FUS by 50% to 80% in the brain and spinal cord.
Shneider administered the drug to the patient it had been designed for.
The patient received repeated infusions of the drug into her spinal canal for 10 months.
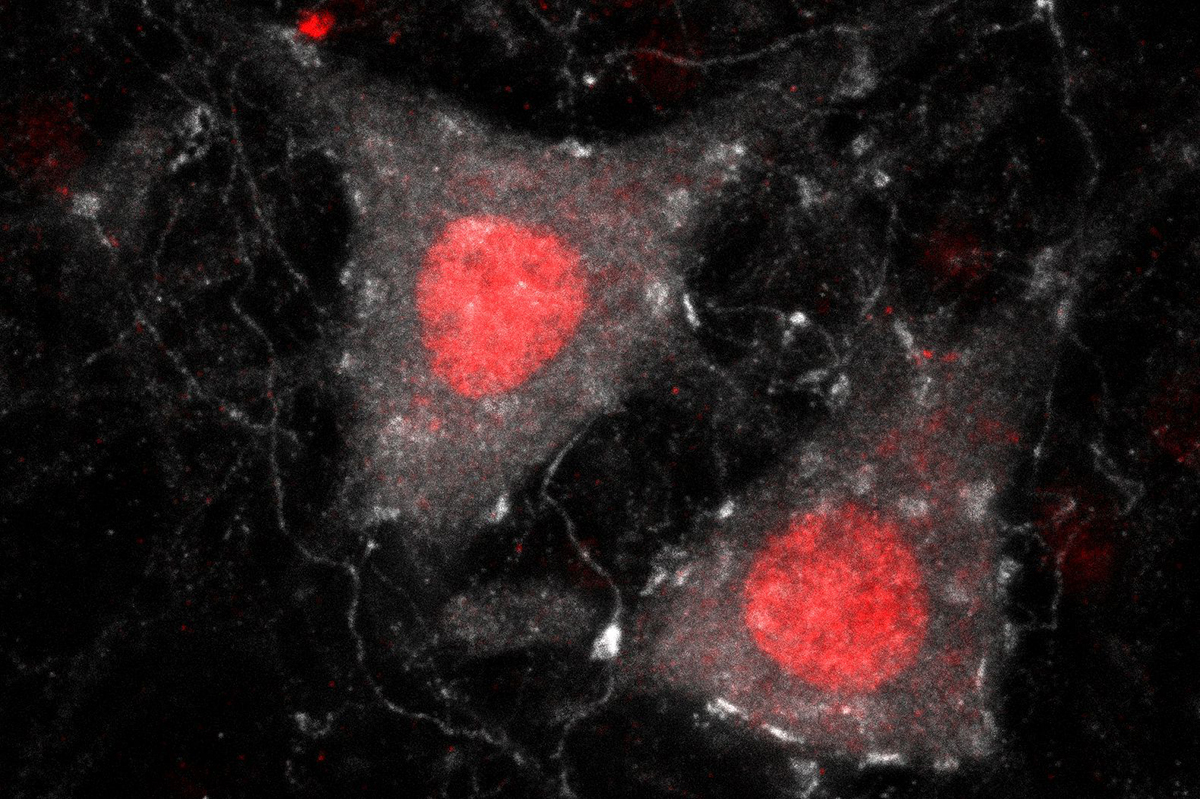
By studying the patient’s brain and spinal cord tissue, researchers found that the drug silenced FUS throughout the nervous system and reversed the toxic nature of FUS and other disease-related proteinsCompared to tissue from untreated FUS-ALS patients and healthy controls, FUS protein aggregates—a pathological hallmark of this form of ALS—were sparse, suggesting that they may have been cleared by the drug
Prior studies in mice suggest that FUS mutations result in the production of an abnormal protein that forms clumps, or aggregates, leading to motor neuron damage
By targeting the faulty gene in a way that suppresses toxic FUS activity, gene silencing products like the antisense oligonucleotide drug could potentially reduce or prevent disease progression
The results were used to support a clinical trial testing the drug in patients with FUS-ALS (NCT04768972)