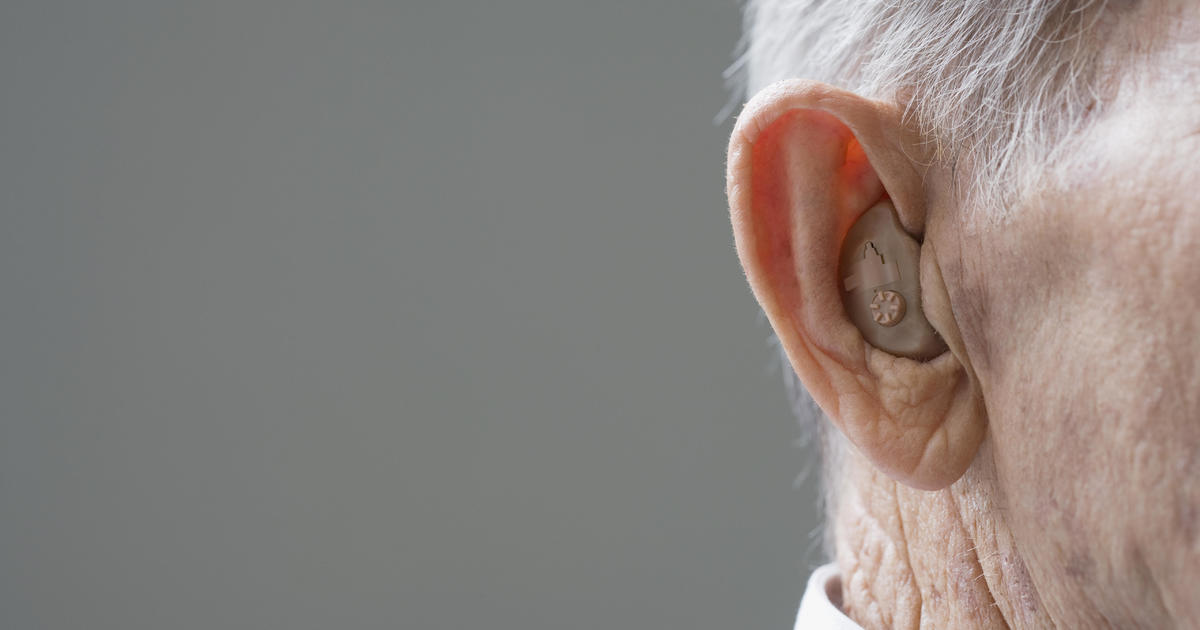
The FDA estimates that nearly 30 million adults could potentially benefit from hearing aids, but only about one-fifth of people with hearing problems use the devices currently.
The new over-the-counter status won't apply to devices for more severe hearing loss, which will remain prescription only.The new rule makes explicit that those devices are not alternatives to FDA-vetted hearing aids.
Tuesday's announcement follows prodding from medical committees and Congress, which in 2017 instructed the agency to lay out a plan for over-the-counter hearing devices.